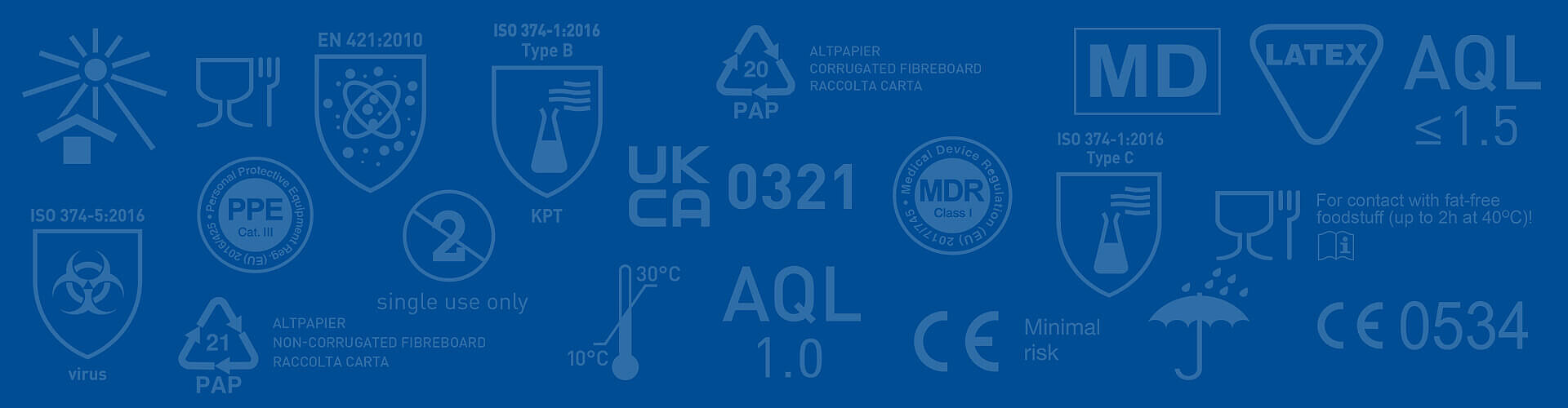
Legislative information about gloves
Everything to know about MDR, PPER and more
Surgical and examination gloves, summarised as medical gloves, are primarily intended to protect the patient from microbial contaminations by healthcare personnel. At the same time, these users are protected from contaminations by the patient they are treating. Thus, all medical gloves must qualify as protective gloves as well.
Personal protective gloves safeguard the hands of professionals in the medical sector, chemical industry, food service, professional cleaning and many other industries. To ensure that gloves successfully do the job of protecting users, they need to comply with a multitude of regulations, which cover different aspects. The regulations and standards are quite detailed and complex, thus can be difficult to comprehend for a non-expert.
As our medical and personal protective gloves are sold and used all around the world, we constantly monitor relevant regulations and standards and how they may affect our customers.
Below are the most recent and important changes affecting the glove industry.
Medical Device Regulation
MDD becomes MDR
The most recent major regulatory change affecting medical device manufacturers is of course the Medical Device Regulation (EU) 2017/745 (“MDR”) and related obligations of economic operators, which has brought new rules for market participants. The MDR, in force since 26 May 2021, aims to create an unprecedented transparency for medical devices.
While the Medical Device Directive 93/42/EEC put the responsibilities of manufacturers in focus, the new MDR expands the spectrum and considers additional market participants. These can be either a manufacturer, an authorised representative, an importer, a distributor or a person placing procedure packs ("kit packs") on the market.
One of the most important changes stemming from the MDR is that companies involved in the supply chain of medical devices must evaluate their role and which new responsibilities arise thereof.
Personal Protective Equipment Regulation
PPE Directive becomes PPE Regulation
In April 2018, PPE Regulation (EU) 2016/425 became applicable and replaced the existing PPE Directive 89/686/EEC. These changes in the regulatory framework for protective gloves entail that requirements for chemical protective gloves were revised and that a new EN ISO 374-1 was published, which in turn led to several changes in the testing and labelling of our products.
EN ISO 21420 - General requirements for protective gloves
EN 420 becomes EN ISO 21410
The well-known standard EN 420 transitioned into EN ISO 21420 without major changes. Previously a European standard, it has now been published as an international standard.
EN ISO 21420 applies to all protective gloves, from thin disposable gloves to thick reusable chemical protection gloves and even to gardening or motorcycle gloves. It defines the general requirements and corresponding test procedures for design principles, glove assembly, harmlessness, comfort and performance, as well as the labelling and information to be provided by the manufacturer.